How can we make enough vaccine for 2 billion people?

Fair access to a vaccine is a vital part of the vaccination process. Image: REUTERS/Agustin Marcarian
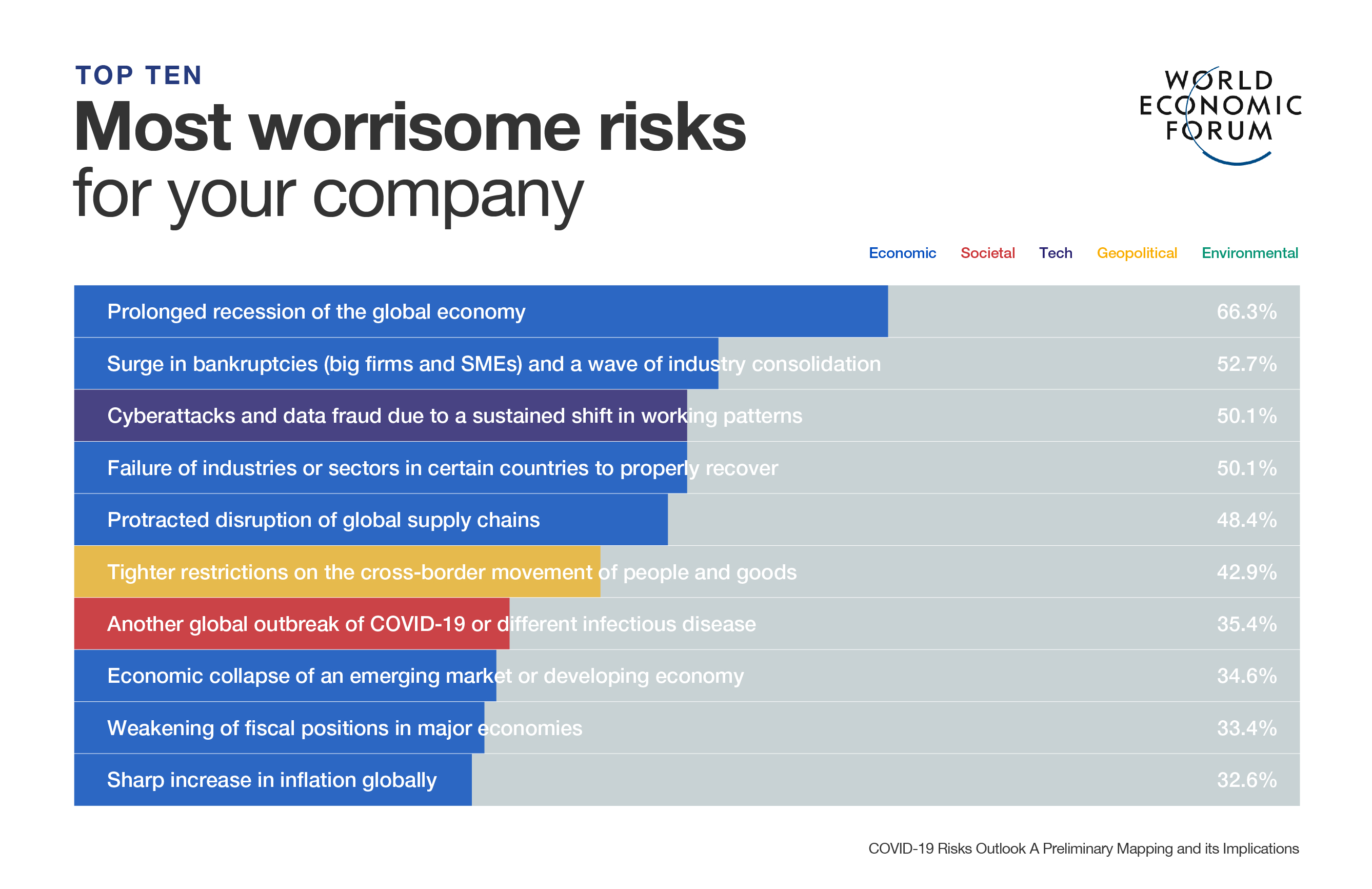
- The World Health Organization and other groups want to make 2 billion doses of vaccine available for the globe’s poorest by late 2021.
- Wealthier nations have already signed deals to secure multiple vaccinations for their populations.
- Making billions of doses will take a global effort, requiring innovation, fast-track approvals and complex supply chains.
With the pandemic, the world's focus has shifted to the creation of an effective vaccine. But once that vaccine is in place, the job of eradicating COVID is far from over. The next challenge will be getting that vaccine to the people who need it most, a project that requires the rapid creation of a vast global supply chain.
One of these plans, COVAX, is a scheme created to provide fast, equitable access to a COVID-19 vaccine around the world. COVAX is trying to raise sufficient funds to make 2 billion doses of effective vaccine available by late 2021.
But that goal could be missed if nations attempt to secure the majority of doses for themselves, which World Health Organization (WHO) Director-General, Tedros Adhanom Ghebreyesus, has called “vaccine nationalism.”
It starts in the lab
The first challenge is to make the vaccine, and it starts small. Around the world, COVID-19 is being grown in cells – kidney cells are commonly used – by teams developing coronavirus vaccines. These are cultured in vats called bioreactors.
Different types of vaccines need different amounts of virus to be produced. For instance, London’s Imperial College, which is working on an RNA-based genetic code vaccine, needs just 1 litre of virus for 200 million people. Conventional vaccines require thousands of litres.
While tiny amounts are needed for vaccine testing (a thousandth of a thousandth of a gram in the case of RNA), for at-risk global populations to be inoculated, vast material will be needed for many of the more than 100 “candidate” vaccine programmes to scale.
Building alliances
“In the early stages of the programme, my team built a consortium with smaller manufacturers in the UK and the Netherlands to go from a three litre scale up to 200 litres,” Sandy Douglas from Oxford’s Jenner Institute tells the BBC. But the team realized they had to get even bigger.
For the Oxford vaccine – among the furthest advanced potential treatments – partnering with pharmaceutical giant AstraZeneca enabled capacity to be increased significantly, adding hundreds of staff to work on the vaccine. Four bioreactors, each with a 2,000-litre capacity, will allow the production of a billion doses of vaccine in two months, the Economist reports.
What is the World Economic Forum doing to manage emerging risks from COVID-19?
However, even AstraZeneca isn’t doing all the work alone. Its partners include the Serum Institute of India, which is preparing to manufacture 1 billion doses of the Oxford vaccine. Other potential vaccines in China and the US are also progressing quickly thanks to complex partnerships.
Working together and innovating
There are currently around 30 candidate vaccines at the more advanced “clinical evaluation” stage and almost 140 more in pre-clinical evaluation, according to the WHO.
Some vaccines could require multiple doses, meaning that potentially 4 billion doses could be needed for the most vulnerable.
Such volume can be more easily reached if companies work together. “No pharma company could make 5 billion doses in the next year or so,” Stéphane Bancel, CEO of biotech company Moderna tells the World Economic Forum’s COVID Action Platform. “But if several of us get to the finish line, then we should be able to put a big dent in the spread of the virus.”
How the vaccine is made can also facilitate fast manufacture at scale. The Oxford trial is employing “single use technologies” – essentially making vaccines in disposable plastic bags – Sandy Douglas tells the BBC. This has made production much easier and less vulnerable to shortages of glassware and other products.
“We need hundreds or thousands of input materials and a shortage of any one of them could derail the whole thing,” says Douglas. To make the supply chain as robust as possible, his team has made sure alternative components can be substituted in if needed.
Delivering where it’s needed most
But making a vaccine in record time will be of limited use if the people who need it most are denied access. This is the concern and motivation behind COVAX, the project led by the WHO, Gavi, the Vaccine Alliance and the Coalition for Epidemic Preparedness Innovations (CEPI).
The programme is designed to pool funds from wealthy nations and nonprofits to develop and distribute a COVID-19 vaccine equitably. It is aiming to deliver 2 billion approved doses by late 2021. The idea is that high-income countries buy vaccines through COVAX and poorer, “funded” countries receive financial assistance.
However, last month the WHO warned that its $2 billion funding target was some way off being met, with around $600 million having been raised and not enough nations signing up.
At the same time, some nations have spent billions of dollars on deals with multiple manufacturers, securing hundreds of millions of doses for their own use.
“We need to prevent vaccine nationalism,” says Tedros Adhanom Ghebreyesus. “As we accelerate the science, solidarity is needed to provide a joint solution to the pandemic.”
Making 2 billion vaccine doses will be an extraordinary achievement; putting them to the best use could be another.
Don't miss any update on this topic
Create a free account and access your personalized content collection with our latest publications and analyses.
License and Republishing
World Economic Forum articles may be republished in accordance with the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International Public License, and in accordance with our Terms of Use.
The views expressed in this article are those of the author alone and not the World Economic Forum.
Stay up to date:
COVID-19
Related topics:
Forum Stories newsletter
Bringing you weekly curated insights and analysis on the global issues that matter.
More on Health and Healthcare SystemsSee all
Mansoor Al Mansoori and Noura Al Ghaithi
November 14, 2025